The Quest for Absolute Zero
Absolute zero is the minimum temperature of matter where the atoms in a substance are rid of any kinetic energy and stops moving. This phenomenon was first theorized back in 1704 by Guillaume Amontons by graphing temperature with pressure. In his experiment of cooling gas, he noticed a decrease in pressure. As the pressure goes down linearly with the decrease in temperature, Amontons believed that had to be a minimum temperature when the pressure is 0 or a vacuum. This led to the hypothesis that there has to be an absolute minimum temperature which matter can achieve. This was the first time anyone thought that there could be a limit of negative temperatures. Amontons could not measure the temperature via his experiment as there were not any instruments able to accurately measure temperatures or in fact, a standard of unit for temperature at that time.
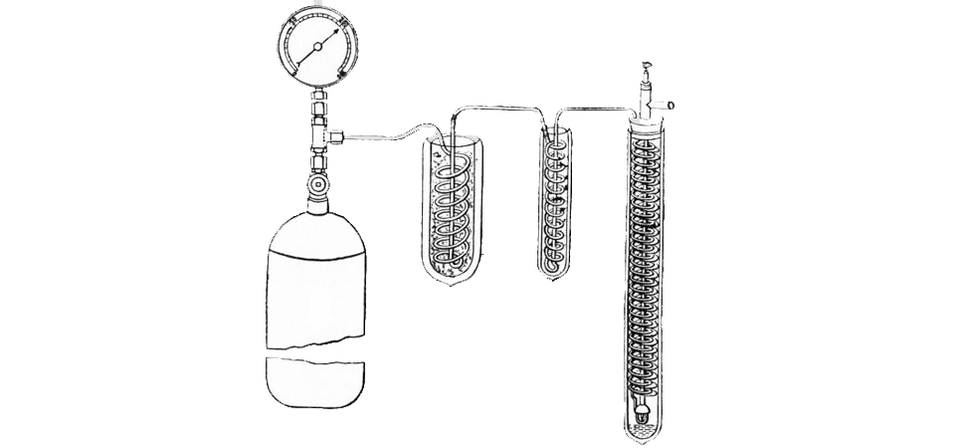
The advancement of accurate instruments soon led to the challenge to liquefy natural gasses. Scientists then were slowly managing to lower the temperature of different substances. James Dewar was one such person in the scientific race to liquefy hydrogen. By increasing the pressure of a gas and suddenly release it, he manages to cool the gas. Based on this, he designed the apparatus to slowly sequentially decrease the temperature of different gasses of decrease temperature to achieve the temperature needed to liquefy hydrogen.
The advancement of accurate instruments soon led to the challenge to liquefy natural gasses. Scientists then were slowly managing to lower the temperature of different substances. James Dewar was one such person in the scientific race to liquefy hydrogen. By increasing the pressure of a gas and suddenly release it, he manages to cool the gas. Based on this, he designed the apparatus to slowly sequentially decrease the temperature of different gasses of decrease temperature to achieve the temperature needed to liquefy hydrogen.
James Dewar’s experimental apparatus was made up of a coil of tubing within another container which contained other liquefied gasses. He placed pressurized gas in a chamber before releasing it to the coil of tubing. The sudden release in pressure and the temperature of the liquid surrounding the coil cools the gas and turns it into a liquid. The chilled liquid is then used as the coolant for the next stage. He continued with this until the gas he cooled was hydrogen. After years of turmoil, he finally succeeded in liquefying hydrogen in 1898 and achieved the lowest temperature of its time -252 degrees Celsius.
The next step down the temperature scale was led by Heike Kamerlingh Onnes. He was determined to liquefy helium at -269 degrees Celsius, shy of absolute zero by 4 degrees. The apparatus used in this experiment was based on the same principle as Dewar’s but was larger and more industrial in nature. It took him many years and many technicians (many who died due to the dangerous nature of his experiments) but he eventually met the challenge to liquefy helium on 10th July 1908. The experiment started around lunch time before finally succeeding several hours later. This monumental achievement was soon awarded the Noble Prize.
The next step down the temperature scale was led by Heike Kamerlingh Onnes. He was determined to liquefy helium at -269 degrees Celsius, shy of absolute zero by 4 degrees. The apparatus used in this experiment was based on the same principle as Dewar’s but was larger and more industrial in nature. It took him many years and many technicians (many who died due to the dangerous nature of his experiments) but he eventually met the challenge to liquefy helium on 10th July 1908. The experiment started around lunch time before finally succeeding several hours later. This monumental achievement was soon awarded the Noble Prize.
The quest for absolute zero also made a large contribution towards many scientific innovations such as super conductivity and the discovery of the Bose–Einstein condensate. These discoveries could only be achieved at low temperatures, only a few billionth degrees from absolute zero.